RESEARCH ARTICLE
Doi: 10.5578/tt.10327
Tuberk Toraks 2016;64(1):34-40

K???k h?creli dışı akciğer kanserinde t?m?r belirteci olarak DR-70'in sensitivite ve spesifitesi
Sibel ARIN?1, Umut Sabri KASAPOĞLU1, ?zlem Makbule AKBAY1, ?zlem ORU?1, Nurcan PAKER2
1 S?reyyapaşa G?ğ?s Hastalıkları ve G?ğ?s Cerrahisi Eğitim ve Araştırma Hastanesi, G?ğ?s Hastalıkları Kliniği,
İstanbul, T?rkiye
1 Clinic of Chest Diseases, Sureyyapasa Chest Diseases and Chest Surgery Training and Research Hospital,
Istanbul, Turkey
2 D?zen Laboratuvarlar Grubu, Klinik Biyokimya, İstanbul, T?rkiye
2 Clinic Biochemistry, Duzen Laboratory Groups, Istanbul, Turkey
?ZET
K???k h?creli dışı akciğer kanserinde t?m?r belirteci olarak DR-70'in sensitivite ve spesifitesi
Giriş: T?m d?nyada kansere bağlı ?l?mlerin en ?nemli nedeni akciğer kanseridir. Akciğer kanserli olgular b?y?k oranda ileri ya da lokal ileri evrede saptanmakta bu nedenle akciğer kanserinin erken tanısı ?ok ?nemlidir. Akciğer kanserinin erken tanısı i?in d?ş?k doz bilgisayarlı tomografi ve t?m?r belirte?leri gibi metodlar ?zerinde durulmaktadır. Bu ?alışmamızda; k???k h?creli dışı akciğer kanserinde bir t?m?r belirteci olarak DR-70'in sensitivitesi ve spesifitesini araştırmayı ama?ladık.
Hastalar ve Metod: Mayıs 2013 ve Nisan 2014 tarihleri arasında k???k h?creli dışı akciğer kanseri tanısı yeni alan 88 olgu ve 86 kronik obstr?ktif akciğer hastalığı tanılı olgu ?alışmaya dahil edilmiştir. Her olgudan serum ?rneği alınıp DR-70 d?zeyi analizi yapılmıştır.
Bulgular: Toplam 174 hasta (152 erkek, 22 kadın) ?alışmaya dahil edildi. Histopatolojik olarak 47 olgu (%53.4') skuam?z h?creli karsinom, 34 olgu (%38.6) adenokarsinom ve 7 olgu (%8) k???k h?creli dışı karsinom idi. Akciğer kanserli olguların ortalama serum DR-70 seviyeleri (2.43 ? 1.82 ?g/mL) kontrol grubunun serum DR-70 seviyelerine (1.15 ? 0.70 ?g/mL) g?re y?ksek saptanmıştır (p< 0.01). DR-70 d?zeyi 1.98 ?g/mL ve ?zerinde olan olgularda akciğer kanseri yakalamada duyarlılık %54.5; ?zg?ll?k %83.7 olarak saptanmıştır. DR-70 d?zeyi 1.98 ?g/mL ve ?zeri olan olgularda hastalık g?r?lme riski 6.171 kat fazladır diyebiliriz.
Sonu?: Fibrin yıkım ?r?nleri d?zeyini ?l?en bir belirte? olan DR-70 y?ksek riskli akciğer kanseri olgularında t?m?r belirteci olarak kullanılabilir.
Anahtar kelimeler: Akciğer kanseri, t?m?r belirteci, duyarlılık, ?zg?ll?k
SUMMARY
The sensitivity and specifity of DR-70 immunoassay as a tumor marker for non-small cell lung cancer
Introduction: Lung cancer is the most important causes of the cancer related mortality. Patients with lung cancer are usually diagnosed at advanced or locally advanced stage, for this reason early diagnosis of lung cancer is very important. For early detection of lung cancer some methods are emphasized such as low-dose computed tomography or tumor biomarkers. In this study we aimed to evaluate DR-70 sensitivity and specificity as a tumor marker in detection of non-small cell lung cancers.
Patients and Methods: Between May 2013 and April 2014, the serum samples from 88 non lung cancer patients, 86 patients with chronic obstructive pulmonary disesase were obtained. Blood samples from each participant were analyzed for DR-70 level.
Results: Totally 174 patients were enrolled to the study (152 male, 22 female). Histopathologically 47(53.4%) patients were diagnosed with squamous cell lung cancer, 34 (38.6%) with adenocarcinoma, and 7 (8%) with non-small cell lung cancer. The mean serum DR-70 levels in lung cancer patients (2.43 ? 1.82 ?g/mL) was significantly higher compared to the 86 non-cancerous subjects (1.15 ? 0.70 ?g/mL) (p< 0.01). DR-70 exhibited clinical sensitivity and specificity of 54.5 and 83.7%, respectively, at an optimal cut off at 1.98 ?g/mL. It could be said that the risk of the presence of the disease is 6.171 times higher in the cases where DR-70 level is 1.98 ?g/mL and higher.
Conclusion: DR-70, a marker used to measure fibrin degradation products, generated by all major cancers, may helps to find high risk lung cancer patients.
Key words: Lung cancer, tumor marker, sensitivity, specificity
Geliş Tarihi/Received: 09.06.2015 • Kabul Ediliş Tarihi/Accepted: 08.10.2015
INTRODUCTION
Today, cancer is one of the most significant health issues. It is also among the primary cause of death in both developed and developing countries (1). All over the world, the most important cause of cancer-related deaths is lung cancer. In 2008, lung cancer has constituted 13% of newly diagnosed cancer cases and 18% of cancer-related deaths (2).
Cases with lung cancer are usually determined in advanced or locally advanced stage. So 70% of cases could not have a chance to be treated surgically, a radical treatment method at the time of diagnosis (3). In spite of the developments in diagnosis and treatments within last 10 years, the ratio of a 5-year-life in lung cancer cases after diagnosis is 15.9% (4).
It is known that the chance to treat the lung cancer cases is closely related with the stage of the disease at the time of diagnosis and there is a target population with a high risk due to smoking and tobacco habit. Therefore, every effort should be made in order to diagnose lung cancer cases at the earliest possible stage (5,6).
AMDL-ELISA DR-70 (FDP) test is the first in vitro diagnostic cancer test approved by? the US Food and Drug Administration (US FDA). DR-70 test measures both fibrin and fibrin degradation products in human serum samples. Fibrin degradation products (FDP) are produced excessively by proteolytic enzymes excreted from cancer cells. It is a proven fact that there is a clear relation between increased FDP levels and cancer detection, its stage, progressionand prognosis (7,8,9). Thus, FDP measurement could be considerably helpful in case of some malign tumours. DR-70 analysis has been previously stated to be effective in the detection of malignancies related with nasopharynx, tongue, gastrointestinal system, breast, ovarium and prostate(10,11,12).
In this study our aim was to detect the sensitivity and specificity of DR-70 immunoassay as a tumour marker in diagnosis of non-small cell lung carcinomas.
MATERIALS and METHODS
Patients and Datas
Between May 2013 and April 2014, at S?reyyapaşa Chest Diseases and Chest Surgery Training and Research Hospital, 88 cases, newly diagnosed with non-small cell lung cancer and 86 cases diagnosed with COPD (Chronic Obstructive Pulmonary Disease)were included into the study.
Absence of an active infection, absence of a history of thrombosis undergone or of a current thrombosis risk, absence of extrapulmonary malignancy history, voluntary participation of patients have been determined as common criteria for the acceptance into the study for the lung cancer group and the COPD control group. At the same time, the cases in the lung cancer group have been conditioned, as a criterion, to be diagnosed with non-small cell lung cancer.
88 cases in the lung cancer group have been evaluated in terms of age, gender, tumoral lesion and its size, tumor histopathology, stage of disease and presence of metastasis. The diagnosis of lung cancer has been verified for all cases by histopathologic examinations. In the staging ofthe disease, TNM 7 lung cancer staging system has been used regarding Stage 1-2 as early stage of a disease, Stage 3-4 as advanced stage of a disease for the cases (13). In the cases of COPD group, age and gender data have been recorded.
A 5 mL sample of peripheral blood was obtained from each subject (both the patients with the non small cell lung carcinoma and the patients with COPD control subjects). The blood was left at room temperaure for 30 minutes, then it was centrifuged at 1300 x g for 10 minutes. Then, all sera were stored at -20?C until they were analysed using DR-70 immunoassay. The serum DR-70 concentration was measured following the manufacturer's protocol for the AMDL-ELISA DR-70 kit (AMDL Inc., Tustin, CA, USA).
The study protocol was approved by local Ethics Committee of a state training and research hospital (Dr. Lutfi Kirdar Kartal Training and Research Hospital, Istanbul-Turkey). Informed consent was signed before patients and healthy individuals were included in this study.
Statistical Analysis
For statistical analyses, NCSS (Number Cruncher Statistical System) 2008 Statistical Software (Utah, USA) program has been used. In addition to descriptive statistical methods (mean, standard deviation, median, ratio, frequency) during the evaluation of work data, Student t test has been employed in the inter-group comparisons of the parameters indicating a normal distribution upon the comparison of quantitative data. In the inter-group comparisons of the parameters not indicating a normal distribution, on the other hand, Mann-Whitney U test has been used. As for the comparison of qualitative data, Chi-Square test has been used while diagnosis screening tests (sensitivity, specificity, PPV, NPV) and ROC curve analysis have been appliedin thecut-off determinationfor parameters. Results have been evaluated within a confidence interval of 95% and significance has been evaluated at a level p< 0.05 and p< 0.01.
RESULTS
Demographics and Clinical Characteristics
of Patients
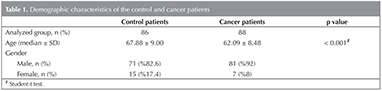
The study has been carried out on 174 patients in total, 12.6% (n= 22) of them being female and 87.4% (n= 152) being male, between May 2013 and April 2014, at Sureyyapasa Chest Diseases and Chest Surgery Training and Research Hospital. The median age of the cases whose ages have ranged from 40 to 86 years, are 64.95 ? 9.19 years. The demographic datas of the groups were shown in Table 1.
While the tumour lesion of 30.7% (n= 27) of the cases with lung cancer who participated in the study has been observed to be the right upper lobe, it has been found to be the right middle lobe for 6.8% (n= 6) of them, the right lower lobe for 25% (n= 22) of them, the left upper lobe for 29.5% (n= 26) of them and the left lower lobe for 8% (n= 4) of them.
Having been examined, the tumour size of the cases with lung cancer being 52.44 ? 25.22 mm on the average has been determined to range from 15 to 134 mm. While the tumour size of 22.7% (n= 20) of the cases has been detected to be 3 cm or less, it has been seen to range from 3 to 7 cm for 53.4% (n= 47) of them and to be bigger than 7 cm for 23.9% (n= 21) of them.
50% (n= 44) of the cases in the patient group have been observed not to be operatedwhereas 44.3% of them (n= 39) have been observed to be subjected to lobectomy and 5.7% of them (n= 5) have been observed to be subjected to pneumonectomy.
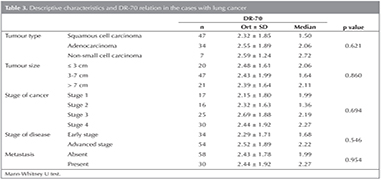
While the type of the tumour for the 53.4% (n= 47) of the cases with lung cancer who participated in the study has been detected to be of squamous cell, it has been observed to be adenocarcinoma for 38.6% of them (n= 34) and non-small cell carcinoma for 8% of them (n= 7).
The cancer stage of 19.3% (n= 17) of the cases with lung cancer are observed to be stage 1, whereas it is observed to be stage 2 for 18.2% of them (n= 16), stage 3 for 28.4% of them (n= 25) and stage 4 for 34.1% of them (n= 30). It is seen that the disease stage of 38.6% of the cases who have participated in the study is early stage, whereas it is observed to be advanced stage for 61.4% of them (n= 54).
While no metastasis is seen in 65.9% (n= 58) of the cases among the lung cancer group, it is observed to exist in 34.1% of them (n= 30). In Table 2, descriptive characteristics of the cases with lung cancer are summarized.
DR-70 Evaluation and ROC Analysis of the Cases of Lung Cancer Group and Control Group
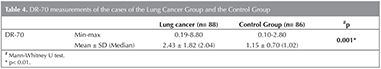
Having ranged from 0.10 to 8.80 ?g/mL, DR-70 measurement of the cases who participated in the study has been determined to be 1.80 ? 1.52 ?g/mL on the average.
A statistically significant difference has not been encountered among the DR-70 measurements of the cases with lung cancer depending on type and size of tumour, stage of disease, existence of metastasis. (p> 0.05) (Table 3).
DR-70 measurements of the cases based on the groups show a statistically significantvariation to a great extent (p< 0.01). Compared to those of the control group, DR-70 measurements of the cases of lung cancer group are significantly higher (Table 4).
Based on this significancy, the determination of a cut-off point for DR-70 has been thought. ROC analysis and diagnosis screening tests have been employed for the determination of a cut-off point for each group.
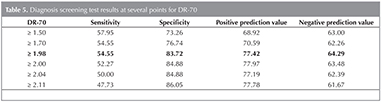
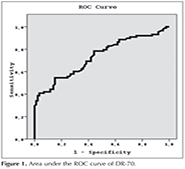
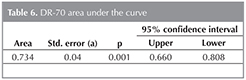
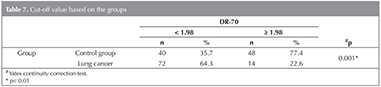
In the cases where DR-70 level is 1.98 ?g/mL and higher, the sensitivity of detecting lung cancer, specificity, positive prediction value and negative prediction value have been determined to be 54.5%, 83.7%, 77.4% and 64.2%,respectively (Table 5). The area under the ROC curve obtained, and standard error have been detected to be 73.4% and 4%, respectively (Figure 1) (Table 6).
A statistically significant relation between the groups and at the cut-off value 1.98 ?g/mL of DR-70 level (p< 0.01) (Table 7). It could be said that the risk of the presence of the disease is 6.171 times higher in the cases where DR-70 levelis 1.98 ?g/mL and higher (ODDS ratio 6.171 [95 % CI: 3.035 - 12.550)].
DISCUSSION
The most crucial point for the control and the prevention of cancer in the world, is the? ability for diagnosing it at the earliest possible stage and then, the reduction of mortality and morbidity through the application of effective treatment methods (14).DR-70 (FDP) test is the first in vitro diagnostic cancer test approved by? the US Food and Drug Administration (US FDA). DR-70 test measures both fibrin and fibrin degradation products in human serum samples. It is a proven fact that there is a clear relation between increased FDP levels and cancer detection, its stage, progression and prognosis (7,8,9). DR-70 analysis has been previously stated to be effective in the detection of malignancies related with nasopharynx, tongue, gastrointestinal system, breast, ovarium and prostate (10,11,12).
In the present study, we evaluate the clinical diagnostic value of DR-70 such as the sensitivity and specificity, positive and negative predictive values in non small cell lung cancers compared to COPD control patiens. The present study demonstrated that the mean serum DR-70 level was higher in non small cell lung cancer patients compared to the COPD patients. DR-70 had a sensitivity of 54.5% and specificity of 83.7% at a cut-off value of 1.98 ?g/mL. Area under the ROC curve (AUC) was 0.734 (95% CI 0.660-0.808, p: 0.001) for the DR-70. The positive predictive value was 77.4%, the negative predictive value was 64.2%.
Wu et al. found that the DR-70 levels increased in lung cancer patients, with an 87% diagnostic sensitivity and a specificity of 95%, was obtained using 335 control subjects and 83 lung cancer patients (15). In a study on 30 subjects with lung cancer whose serum DR-70 cut-off values were measured as 1.0 ?/mL and a healthy control group of 85 subjects, sensitivity and specificity have been found to be 87% and 95%, respectively (16). In a study performed by Adonis et al. on a population with a high risk of lung cancer, the sensitivity of serum DR-70 was found to be 90% (17). In our study, the sensitivity of DR-70 in the cases of non-small cell lung cancer has been measured as 54.5% and we attribute it to the fact that the control group consisted of the cases with COPD.
Fields et al. found that mean DR-70 level of cancer patients were 3 times higher compared to the control subjects (18). The results of the present study were? consistent with the results. In addition to that, the mean DR-70 level in the cases of non-small cell lung cancer has been found to be 2 times higher than that of the control group.
Kerber et al. have determined that DR-70 levels in advanced gastrointestinal cancers increase (19). In this sense, a positive correlation has been established between high DR-70 level and tumour burden and the number of metastasis. In another study, on the other hand, no correlation has been detected between high DR-70 level and histological type of tumour and stage of disease (20). The present study serum DR-70 levels were not correlated with the histological subtype of the tumor and tumor stage.
This study does have several limitations. For example we did not include small cell lung cancer patients and healthy controls in the sample population. The patient sample size that is 174 cases may not be sufficiently large to determine the cut off level between non small cell lung cancer patients and COPD controls.
CONCLUSION
We are in the opinion that comprehensive studies including high risk phenomenon and healthy ones in terms of lung cancer in particular in our country are required for DR-70 which has been indicated in the previous studies that it has high sensitivity and specificity in cancer screening.
REFERENCES
- World Health Organization. The global burden of disease: 2004 Update. Geneva: World Health Organization, 2008.
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin 2011;61:69-90.
- Spiro SG, Porter JC. Lung cancer-Where are we today? Current advances in staging and non surgical treatment. Am J Respir Crit Care Med 2002;166:1166-96.
- Ettinger DS. Ten years of progress in non-small cell lung cancer. J Natl Compr Canc Netw 2012;10:292-5.
- Mountain CF. Revisions in the International System for Staging Lung Cancer. Chest 1997;111:1710-7.
- Alberg AJ, Samet JM. Epidemiology of lung cancer. Chest 2003;123(Suppl 1);S21-S49.
- Ossowski L, Quigley JP, Kellerman GM, Reich E. Fibrinolysisassociated with oncogenic transformation. Requirement of plasminogen for correlated changes in cellular morphology, colony formation in agar, and cell migration. J Exp Med 1973;138:1056-64.
- Gerner C, Steinkellner W, Holzmann K, Gsur A, Grimm R, Ensinger C, et al. Elevated plasma levels of crosslinked fibrinogen gamma-chain dimer indicate cancer-related fibrin deposition and fibrinolysis. Thromb Haemost? 2001;85:494-501.
- Perry R, Sheila MA, Barbara B. Elevated fibrinogen-fibrin degradation products (FDP) in serum of colorectal cancer patients. Anal Lett 2004;37:2965-76.
- Small-Howard AL, Harris H. Advanteges of the AMDL-ELISA DR-70 (FDP) assay over carcinoembryogenic antigen (CEA) for monitoring colorectal cancer patients. J. Immunoassay Immunochem 2010;31:131-47.
- Wu D, Zhou X, Yang G, Xie Y, Hu M, Wu Z, et al. Clinical performance of the AMDL DR-70(TM) immunoassay kit for cancer detection. J Immunoassay 1998;19:63-72.
- Li X, Qaio Z, Long X, Wei J, Cheng Y. Serum concentration of AMDL DR-70 for the diagnosis and prognosis of carcinoma of the tongue. Br J Oral Maxillofac Surg 2005;43:513-5.
- Rami-Porta R, Ball D, Crowley J, Giroux DJ, Jett J, Travis WD et al; International Staging Committee; Cancer Research and Biostatistics; Observers to the Committee;Participating Institutions. The IASLC Lung Cancer Staging Project: Proposals For The Revision Of The T Descriptors İn The Forthcoming (Seventh) Edition Of The TNM Classification For Lung Cancer. Thorac Oncol 2007;2:593-602.
- Srinivas PR, Kramer BS, Srivastava S. Trends in biomarker research for cancer detection. Lancet Oncol 2001;2:698-704.
- Wu DF, Zhou X, Anderson G, Fuentes A, Slater LM, Narinesingh D, et al. Sensitivity and specificity of DR-70 lung cancer immunoassay. Anal Lett 1999;32:1351-62.
- Motamed-Khorasani A, Grimes R, Weber DF. The validation of DR-70 efficiency in early detection of lung cancer. Lung Cancer 2011;71(Suppl 2):S29-S30.
- Adonis M, D?az J, Urz?a U, Chahuan M, Avaria P, Miranda R, et al. Detection of preneoplastic lesions using biological and genemic lung cancer biomarkers in a high risk Chilean population. J Thorac Oncol 2011;6(Suppl 2):S980-S1.
- Fields A, Poppema S, Jha N, Marcushamer S, McNamee C, Hanson J, et al. Serum levels of circulating extracellular matrix complex (CEMC) in lung cancer patients: potential use as a tumor marker. 12th International Conference on Human Tumor Markers, 1995:11-14.
- Kerber A, Trojan J, Herrlinger K, Zgouras D, Caspary WF, Braden B. The new DR 70 immunoassay detects cancer of the gastrointestinal tract: a validation study. Aliment Pharmacol Ther 2004;20:983-7.
- Sengupta A, Saha K, Jash D, Banerjee SN, Biswas NM, Dey A. Role of DR-70 immunoassay in suspected malignant pleural effusion. Lung India 2013;30:321-6.
Yazışma Adresi (Address for Correspondence)
Dr. Umut Sabri KASAPOĞLU
S?reyyapaşa G?ğ?s Hastalıkları ve
G?ğ?s Cerrahisi Eğitim ve Araştırma Hastanesi,
G?ğ?s Hastalıkları Kliniği,
İSTANBUL - TURKEY
e-mail: umutkasapoglu@gmail.com