SHORT REPORT
Doi: 10.5578/tt.8468
Tuberk Toraks 2015;63(1):37-41

Nadir g?r?len bir dif?z parankimal akci?er hastal???: Dif?z pulmoner meningotelyomatozis
Nazan ?EN1, Emine Tuba CANPOLAT2, Zafer KO?3
1 Ba?kent ?niversitesi T?p Fak?ltesi, Adana Uygulama ve Ara?t?rma Merkezi, G???s Hastal?klar? B?l?m?, Adana, T?rkiye
1 Department of Chest Diseases, Faculty of Medicine, Baskent University, Adana Practice and Research Center,
Adana, Turkey
2 Ba?kent ?niversitesi T?p Fak?ltesi, Adana Uygulama ve Ara?t?rma Merkezi, Patoloji B?l?m?, Adana, T?rkiye
2 Department of Pathology, Faculty of Medicine, Baskent University, Adana Practice and Research Center, Adana, Turkey
3 Ba?kent ?niversitesi T?p Fak?ltesi, Adana Uygulama ve Ara?t?rma Merkezi, Radyoloji B?l?m?, Adana, T?rkiye
3 Department of Radiology, Faculty of Medicine, Baskent University, Adana Practice and Research Center, Adana, Turkey
?ZET
Nadir g?r?len bir dif?z parankimal akci?er hastal???: Dif?z pulmoner meningotelyomatozis
Pulmoner meningotelyal nod?ller (PMN) s?kl?kla rezeke edilen akci?er parankim ?rneklerinin ve otopsilerin patolojik incelenmesi s?ras?nda rastlant?sal olarak saptan?r. Bu nod?ller genellikle asemptomatik ve ?o?u zaman tektir. Dif?z pulmoner tutulum s?kl??? daha azd?r. PMN benign lezyonlar olup, neoplastik ve neolastik olmayan akci?er hastal?klar? ve nadiren ekstrapulmoner hastal?klarla ili?kili bulunmu?tur. Multipl bilateral pulmoner nod?lleri olan ve video e?li?inde torakoskopik cerrahi (VATS) ile "dif?z pulmoner meningotelyomatozis" tan?s? alan bir kad?n hasta sunulmu?tur. Dif?z pulmoner meningotelyomatozis radyolojik olarak dif?z bilateral akci?er nod?lleri olan hastalarda ay?r?c? tan? i?inde yer almal?d?r.
Anahtar kelimeler: Pulmoner meningotelyal nod?ller, meningotelyomatozis, mikronod?ller
SUMMARY
A rarely seen diffuse parenchymal lung disease: diffuse pulmonary meningotheliomatosis
Pulmonary meningothelial-like nodules (MLNs) are usually detected incidentally during pathologic evaluation of resected pulmonary parenchymal specimens and autopsies. These nodules are generally asymptomatic and most often single. Diffuse pulmonary involvement by MLNs is less frequently described. MLNs are benign lesions and have been associated with neoplastic and non-neoplastic pulmonary conditions and occasionally with extrapulmonary diseases. We report a case of a female patient presenting with multiple and bilateral pulmonary nodules diagnosed with "diffuse pulmonary meningotheliomatosis" by video-assisted thoracoscopic surgery (VATS). Diffuse pulmonary meningotheliomatosis should be included in the differential diagnosis of diffuse bilateral lung nodules in the radiologic studies.
Key words: Minute pulmonary meningothelial-like nodules, meningotheliomatosis, micronodules
Geli? Tarihi/Received: 19.10.2014 • Kabul Edili? Tarihi/Accepted: 25.10.2014
INTRODUCTION
Pulmonary meningothelial-like nodules (MLNs) are rarely seen lesions consisted of small epitheloid cell clusters and are located in the parenchyma of the lungs. They were previously known as pulmonary chemodectoma (1). The patient population shows a marked female predominance (1,2,3). Although they usually appear as solitary nodules, multiple lesions with unilateral involvement have also been defined (1,4). Diffuse bilateral meningothelial nodules, which resemble metastatic disease or interstitial lung disease, are extremely rare (1,5). The term ?meningotheliomatosis? has been suggested for these lesions (1). The present case, diagnosed with "diffuse pulmonary meningtheliomatosis", was presented for to highlight the necessity of considering this condition in the differential diagnosis of diffuse parenchymal nodular lesions.
CASE REPORT
A 66-year-old female patient presented with non-productive cough. She had 13 pack/year smoking history. The physical examination of the patient was within normal limits. Her past medical history was notable for diabetes mellitus, hypertension and chronic renal disease. She had no known malignancy. On her laboratory analysis, BUN was 38 mg/dL, creatinine was 2.45 mg/dL, and blood glucose was 167 mg/dL; results of other blood and urine analyses were within the normal ranges.
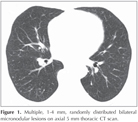
The patient showed normal spirometry and carbon monoxide diffusion capacity, but slightly decreased total lung capacity (71%). Current computed tomography (CT) of the thorax at the time of admittance displayed diffuse, multiple, randomly distributed 1-4 mm nodular lesions in the parenchyma of both lungs (Figure 1). The patient had been followed by thoracic CT scan in another center and there had been no radiological difference for one and a half year period. She had been referred to our hospital for definitive diagnosis. As the nodules were small, have no bronchus sign and revealed no endobronchial lesion on thoracic CT scan she underwent video-assisted thoracoscopic surgery (VATS) for its higher diagnostic yield than other conventional diagnostic procedures such as bronchoscopy or transthoracic needle biopsy. Nodular configurations, which were also seen on visceral pleural surface, were observed during surgical operation.
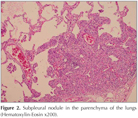
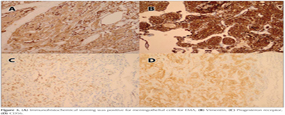
Histopathological examination revealed small, 1 to 4 mm nodular lesions consisted of clusters of multiple cells with round-to-oval nucleus and large cytoplasm (Figure 2). While these cells were immunohistochemically positive for EMA, vimentin, progesterone receptor (PR) and CD56, they were negative for keratin, smooth muscle actin, chromogranine, CD34 and sinaptophysin (Figure 3A-D). The patient, whose histopathological findings were reported as ?meningothelial nodules?, was diagnosed with "diffuse pulmonary meningtheliomatosis" based on her radiological findings and no underlying lung disease was identified.
DISCUSSION
MLNs were first defined by Korn et al. in 1960 (6). Although it has been more than 50 years, its etiology and pathogenesis remain uncertain (1,4,5). It has similar histological, ultrastructural and immunohistochemical features with meningiomas (4). Along with the use of immunohistochemistry, evidence has been obtained for meningothelial origin while confirming the absence of neuroendocrine differentiation in MLNs (2). Immunohistochemical studies of the cells showed strong positivity for progesterone receptor, EMA, vimentin and CD56 but negative staining for cytokeratin, actin, S-100 protein, CD34, chromogranine, synaptophysin and neuron-specific enolase (2). EMA and vimentin are closely associated with meningiomas (1). Although CD56 is frequently encountered in neuroendocrine proliferation, it has been reported also in meningiomas. Therefore, CD56 staining in PMLNs supports menigothelial differentiation (2). Mukhopadhyay et al. detected patchy staining with CD56 in 50-100% of the cells (2). Staining pattern has been reported to be cytoplasmic and membranous and mild-to-moderate in intensity. In the present case, staining with CD56 was detected in more than 70% of the cells and the staining pattern was observed to be cytoplasmic and membranous and moderate in intensity. Niho et al. performed immunohistochemical and clonal analyses and concluded that MLN is a reactive proliferation rather than a neoplastic event (7). Mutational analysis of MLNs and intracranial meningiomas suggested that these are two unrelated conditions and that MLNs represent a reactive event (4). Despite all these observations, presence of meningothel in the lungs and the relation between underlying pulmonary disease and MLNs remain unclear (2).
Incidence of MLNs in surgical lung biopsies is higher than the incidence reported in autopsy studies? (13.8% vs. 1.1%) (2,8). The authors postulated that such a difference in its incidence might be associated with presence of an underlying lung disease in all surgical lung biopsies raising the thought that MLNs are associated with chronic lung diseases (2). The highest incidence of MLNs has been noted in patients with pulmonary thromboembolism (42%). Association with smoking-induced interstitial lung disease and neoplasia, particularly lung adenocarcinoma, has been documented as well. The low incidence of MLNs (4%) in acute lung injury suggests that chronicity may be important for the formation of MLNs (2). Mizutani et al. found MLNs to be higher in malignant pulmonary tumors than in benign diseases (7.3% and 2.5% respectively, p= 0.044) (3). It has been reported to be more prevalent particularly in the patients with adenocarcinoma versus the patients with other primary lung cancers (9.4% vs. 4.5%, p< 0.01). No underlying neoplastic or non-neoplastic pulmonary disease was detected in the present case.
MLNs is more frequently seen in females and in the 6th and 7th decades of life (2,6). Male-female ratio is found to be 2.3:1. Such a female predominance was considered to be associated with the presence of progesterone receptor immunoreactivity. The absence of MLNs in pediatric lung specimens indicates that they are not congenital (2).
The etiology of multiple pulmonary nodules (MPNs) is complex. MPNs may be caused by malignant and benign diseases. MPNs that are < 5 mm in diameter are more likely to be benign lesions; however, they are more likely to be malignant than benign in patients with a known malignant primary tumor. Benign diseases that are known to cause MPNs include infections, non-infectious inflammatory conditions, and pneumoconioses. In asymptomatic patients, further considerations include the presence of rheumatoid nodules, amyloidosis, and arteriovenous malformations. Hamartomas and smooth muscle tumors such as leiomyomas are other less common conditions (9). An inflammatory condition or a benign neoplasm was considered in the differential diagnosis of our patient. VATS, is a technique that has recently become available for evaluating pulmonary nodules, was performed to obtain a definitive diagnosis for its high diagnostic yield in the present case.
Although MLNs usually appears as a solitary lesion, multiple lesions have been identified in 41% of the patients. Diffuse bilateral pulmonary involvement by MLNs is termed as ?diffuse pulmonary meningotheliomatosis?. In a molecular study multiple lesions have demonstrated to show a greater genetic instability along with more frequent loss of heterozygosity as compared to solitary lesions. Therefore, multiple and diffuse lesions are likely to represent a transition stage between reactive and neoplastic condition (4).
There is limited number of radiological studies concerning radiological findings of MLNs. Thoracic CT scan of the present case demonstrated randomly distributed sharply circumscribed multiple micronodules. Radiologically, MLNs are usually detected as diffuse, multiple nodules in both lungs with the size ranging between 100 ?m-3 mm (1,5,10,11,12,13,14). They may be seen as micronodules (11), reticulonodular infiltrations (1), or ground-glass opacities (10,12,13). Presence of nodules with cavitation has been reported in some cases (5,14). Lee et al detected a 11.3 mm nodule and bilateral micronodules in both lungs on thoracic CT scan of a 52-year-old man (15). The size of MLNs in this report were larger than described in other studies (15).
In conclusion; the present case, of which histopathological examination revealed ?meningothelial-like nodules?, was diagnosed with ?diffuse pulmonary meningotheliomatosis? based on her radiological findings. It can mimic metastatic disease particularly in patients with malignancy and may cause misstaging. The present case has been presented to highlight the necessity of considering this disease in the presence of undefined diffuse bilateral parenchymal nodular lesions.
CONFLICT of INTEREST
None declared.
REFERENCES
- Suster S, Moran CA. Diffuse pulmonary meningotheliomatosis. Am J Surg Pathol 2007;31:624-31.
- Mukhopadhyay S, El-Zammar OA, Katzenstein AA. Pulmonary meningothelial-like nodules. New insights into a comman but poorly understood entity. Am J Surg Pathol 2009;33:487-95.
- Mizutani E, Tsuta K, Maeshima AM, Asamura H, Matsuno Y. Minute pulmonary meningothelial-like nodules: clinicopathologic analysis of 121 patients. Human Pathol 2009;40:678-82.
- Ionescu DN, Sasatomi E, Aldeeb D, Omalu BI, Finkelstein SD, Swalsky PA, et al. Pulmonary meningothelial-like nodules. A genotypic comparison with meningiomas. Am J Surg Pathol 2004;28:207-14.
- Kraushaar G, Ajlan AM, English JC, M?ller NL. Minute pulmonary meningothelial-like nodules: a case of incidentally detected diffuse cystic micronodules on thin-section computed tomography. J Comput Assist Tomogr 2010;34:780-82.
- Korn D, Bensch K, Liebow A, Castleman B. Multiple minute pulmonary tumors resembling chemodectomas. Am J Pathol 1960;37:641-72.
- Niho S, Yokose T, Nishiwaki Y, Mukai K. Immunohistochemical and clonal analysis of minute pulmonary meningothelial-like nodules. Hum Pathol 1999;30:425-29.
- Pelosi G, Maffini F, Decarli N, Viale G. Progesterone receptor immunoreactivity in minute meningotheloid nodules of the lung. Virchows Arch 2002;440:543-46.
- Beigelman-Aubry C, Hill C, Grenier PA. Management of an incidentally discovered pulmonary nodule. Eur Radiol 2007;17:449-66.
- Kuroki M, Nakata H, Masuda T, Hashiguchi N, Tamura S, Nabeshima K, et al. Minute pulmonary meningothelial-like nodules: High resolution computed tomography and pathologic correlations. J Thorac Imaging 2002;17:227-29.
- Sellami D, Gotway MB, Hanks DK, Webb WR. Minute pulmonary meningothelial-like nodules: thin-section CT appearance. J Comput Assist Tomogr 2001;25:311-13.
- Kfoury H, Arafah MA, Arafah MM, Alnassar S, Hajjar W. Mimicry of minute pulmonary meningothelial-like nodules to metastatic deposits in a patient with infiltrating lobular carcinoma: a case report and review of the literature. Korean J Pathol 2012;46:87-91.
- Kamiya K, Yoshizu A, Kashizaki F, Hida N, Hayashi H. Minute pulmonary meningothelial-like nodules coexisting with pulmonary cryptococcosis mimicking lung cancer. Gen Thorac Cardiovasc Surg 2013;61:659-62.
- Mora RB, Nieto JMS, Hu C, Mateos EA, Bascunana AG, Rodriguez MR. Diffuse pulmonary meningotheliomatosis diagnosed by transbronchial lung biopsy. Respiration 2013;86:145-48.
- Lee SK, Kim GJ, Kim YJ, Leem AY, Hwang ED, Kim SK, et al. Minute pulmonary meningothelial-like nodules simulating hematogenous lung metastasis: a case report. Tuberc Respir Dis 2013;75:67-70.
Yaz??ma Adresi (Address for Correspondence)
Dr. Emine Tuba CANPOLAT
Ba?kent ?niversitesi T?p Fak?ltesi,
Adana Uygulama ve Ara?t?rma Merkezi,
Dadalo?lu Mah 39 Sok. No: 6,
Y?re?ir, ADANA - TURKEY
e-mail: nazansen68@gmail.com