?ntravezikal Bacillus Calmette-Guerin imm?noterapisi sonras? geli?en miliyer t?berk?loz
Abdullah ??M?EK, M?jgan G?LER, Sibel G?NAY, Nermin ?APAN
Atat?rk G???s Hastal?klar? ve G???s Cerrahisi E?itim ve Ara?t?rma Hastanesi, G???s Hastal?klar? Klini?i, Ankara.
Tuberk Toraks 2013; 61(3): 252-254 • doi:10.5578/tt.4692
Geli? Tarihi/Received: 11/01/2013 - Kabul Edili? Tarihi/Accepted: 31/01/2013
Intravesical administration of the Bacillus Calmette-Guerin (BCG) is an effective treatment for superficial carcinoma of bladder (1). Complications of such immunotherapy are usually local, but may result in severe and systemic manifestations (2). Miliary tuberculosis is a very rare complications of BCG immunotherapy that is associated to high mortality.
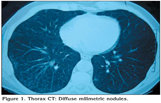
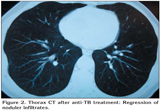
A nonimmunocompromised 56 year old man had undergone transurethral resection (TUR) for low-grade superficial papillary urothelial bladder cancer. Then, six weekly instillations of intravesical BCG, a Pasteur strain, 120 mg each had been planned for treatment. Seven days after TUR, first intravesical BCG instillation administered. He had developed 40?C fever one day after that instillation. Thorax computed tomography (CT) had revealed diffuse multiple milimetric nodules especially at the lower lobes of the both lungs (Figure 1). Sputum smears acide fast bacillus (AFB) and tuberculosis cultures were found to be negative for six times. In flexible bronchoscopy, secretions were seen coming from basal segments of right lower lobe. Bronchoalveolar lavage (BAL) and transbronchial biopsy specimen of lung tissue was taken from posterior basal segment of right lower lobe. Tissue biopsy revealed a granulomatous inflammation with caseation necrosis. Tissue AFB, tuberculosis cultures, tuberculosis polymerase chain reaction (PCR) were negative. BAL AFB and tuberculosis PCR were negative also. Hemoglobine level decreased to 9.1 g/dL, leukocyte count decreased to 3070 ?L. AST and ALT levels increased to 95 U/L and 69 U/L respectively. He had been considered as miliary tuberculosis. Antituberculosis treatment with HRZE had started and corticosteroid treatments had been given for seven days. Abdominal ultrasonography had revealed hepatosplenomegaly. Serologic markers for hepatitis were negative. Five months after antituberculosis treatment, thorax intravesical showed marked regression of noduler infiltrates (Figure 2). Leukocyte count, hemoglobine level became normal. Cultures of BAL and sputum smears were negative for Mycobacterium tuberculosis. Antituberculosis treatment was given 12 months.
Systemic side effects of intravesical BCG immunotheraphy occur in %5 of patients, varying from mild malaise and fever to, in rare cases, life threatening or fatal sepsis (1). Lung involvement can appear as diffuse reticulonodular opacities, a miliary or alveolar pattern, or pleural effusion (3,4,5).
Currently it is recommended to start intravesical BCG 120 mg per instillation session at least after 7-14 days of tumor resection or until postoperative bleeding is over (6). A systemic BCG reaction is much more likely to occur if BCG is administered within one week of either TUR or traumatic bladder catheterisation (7). In this case, we think that the reason of disease was early intravesical administration of BCG (instillation seven days after TUR).
The pathogenic mechanism of pulmonary toxicity is controversial; although some authors favor a hypersensitivity response, others suggest a dissemination of infection (3,8). Our case had developed disease one day after first BCG instillation. Both in the hypersensitivity response and dissemination of infection, disease can develop so early. In some studies, AFB was found negative, but in others, AFB was found positive in postbronchoscopic sputum and lung tissue and AFB culture was positive in lung tissue (3,4,9). In our case, finding sputum smears, BAL and lung tissue negative for AFB, tuberculosis cultures and tuberculosis PCR suggest a hypersensitivity response as a mechanism. But hepatosplenomegaly, high level of liver enzymes and decrease in leukocyte count and hemoglobine level may show haematogenous spread of M. tuberculosis. Indirect evidence to support disseminated BCG infection were the miliary pattern in thorax CT, the pulmonary caseating granuloma formation and haematogenous spread of disease.
The treatment remains controversial. Present case was treated with antituberculosis drugs for 12 months and corticosteroid given for one week. We think that in the suspicious of miliary tuberculosis after intravesical BCG administration, combination of antituberculosis and corticosteroid treatments must be started.
Our case had developed miliary tuberculosis after first BCG instillation. In Rabe's, Palayew's, Foster's studies miliary tuberculosis developed after 16th BCG instillation, after 5th instillation and after 7th treatment respectively but in Erg?n's study it was developed after first BCG administration (4,5,9,10). What are the reasons of differences in time miliary tuberculosis started in? Perhaps these are due to severity of urethral trauma caused by operation, first BCG instillation time, BCG dosage and patient's immunity.
In conclusion, miliary tuberculosis can develope in any time after starting intravesical BCG treatment. If a fever developes after intravesical BCG immunotherapy, miliary tuberculosis should be kept in mind and chest X-ray should be performed. Because of high mortality, antituberculosis and corticosteroid treatments should be initiated immediately even if sputum smears are negative for AFB. Disseminated BCG infection is more likely to be the mechanism for the systemic complications of intravesical BCG.
CONFLICT of INTEREST
None declared.
REFERENCES
- Lamm DL, van der Meijden PM, Morales A, Brosman SA, Catalona WJ, Herr HW, et al. Incidence and treatment of complications of bacillus Calmette-Guerin intravesical therapy in superficial bladder cancer. J Urol 1992; 147: 596-600.
- Lamm DL, Steg A, Boccon-Gibod L, Morales A, Hanna MG Jr, Pagano F, et al. Complications of Bacillus Calmette-Guerin immunotherapy: review of 2602 patients and comparison of chemotherapy complications. Prog Clin Biol Res 1989; 310: 335-55.
- McParland C, Cotton DJ, Gowda KS, Hoeppner VH, Martin WT, Weckworth PF. Miliary Mycobacterium bovis induced by intravesical bacille Calmette-Gu?rin immunotherapy. Am Rev Respir Dis 1992; 146: 1330-3.
- Rabe J, Neff KW, Lehmann KJ, Mechtersheimer U, Georgi M. Miliary tuberculosis after intravesical bacille Calmette-Gu?rin immunotherapy for carcinoma of the bladder. Am J Roentgenol 1999; 172: 748-50.
- Erg?n P, Y?lmaz Turay ?, Erdo?an Y, Oran D, Biber ?, Demira? F. Miliary tuberculosis induced by intravesical Bacillus Calmette-Gu?rin immunotherapy: a rare complication. Turkish Respiratory Journal 2003; 4: 79-81.
- Manzanilla-Garc?a HA, Maldonado-?vila M, Aguilar-Barradas J, et al. Bacillus Calmette-Gu?rin immunotherapy in the treatment of superficial bladder cancer. Rev Mex Urol 2009; 69: 307-13.
- Meyer JP, Persad R, Gillatt DA. Use of bacille Calmette-Gu?rin in superficial bladder cancer. Postgrad Med J 2002; 78: 449-54.
- Israel-Biet D, Venet A, Sandron D, Ziza JM, Chretien J. Pulmonary complications of intravesical Bacille Calmette-Gu?rin immunotherapy. Am Rev Respir Dis 1987; 135: 763-5.
- Palayew M, Briedis D, Libman M, Michel RP, Levy RD. Disseminated infection after intravesical BCG immunotherapy. Detection of organisms in pulmonary tissue. Chest 1993; 104: 307-9.
- Foster DR. Miliary tuberculosis following intravesical BCG treatment. Br J Radiol 1997; 70: 429.
Yaz??ma Adresi (Address for Correspondence):
Dr. Abdullah ??M?EK,
Atat?rk G???s Hastal?klar? ve
G???s Cerrahisi E?itim ve Ara?t?rma Hastanesi,
G???s Hastal?klar? Klini?i, ANKARA - T?RK?YE
e-mail: abdullahsimsek1@yahoo.com.tr